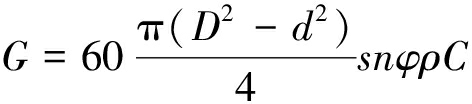淖毛湖煤和棉秆共热解协同效应分析
0 引 言
新疆地区煤炭资源储量丰富,约占全国40%,位居全国首位[1]。其中,吐哈地区煤炭普遍挥发分高,适合用中低温热解方式制备高附加值焦油、煤气和半焦[2-3]。焦油可提取苯类、酚类、萘类和蒽类等化合物,生产合成纤维、染料和香料等;煤气可用作燃料气或化工原料;半焦可用作配煤添加剂、电极原料、气化原料、活性炭和高炉喷吹燃料等[4]。因此煤炭分级分质利用方式符合双碳背景下煤炭转化技术的发展方向和趋势。
然而,煤炭缺氢特性限制了热解的转化率,影响了热解系统的稳定性。煤单独热解时存在焦油产率低、焦油中重质组分(沸点≥360 ℃)含量高、油尘分离困难等问题,这将导致下游装置的频繁堵塞[5-6]。研究发现氢气气氛可以有效稳定煤热解产生的自由基碎片,提高热的解焦油产率和品质[7]。但氢气价格昂贵,经济效益差,因此寻找一种廉价易得的氢源是至关重要的。目前富氢废弃物如生物质[8]、塑料[9]、石油残渣[10]和轮胎[11]等被认为是氢气的替代品。其中,生物质因为储量大、可再生、CO2近零排放和H/C比高等特点[12-13]被广泛地关注,可在共热解中充当供氢体,为煤热解提供氢源。
热解遵循自由基反应机理,共热解同样如此[14]。共热解时,生物质和煤共价键相继断裂并产生自由基碎片,自由基碎片进一步结合,生成焦油、气体和半焦[15-16]。但二者共热解协同作用受诸多因素影响,如反应器类型、热解温度、升温速率、填料方式、混合比例等[17-18]。其中,填料方式是影响共热解产物分布和协同效应的主要因素之一。LI等[19]研究进料混合方式对白音花褐煤和松木共热解产物分布和焦油组成的影响。结果表明煤层在松木层上层时,共热解协同效应好。与计算值相比,焦油产率增加8.29%。且在该进料方式下焦油中烷基愈创木酚相对含量最高,为40.12%。ZHU等[20]考察3种混合方式对淖毛湖褐煤与和柏木共热解产物的影响。发现采用煤在上层、柏木在下层的装填方式时焦油产率更高,达44.68%;焦油中3~4环芳香化合物含量增加,导致焦油品质变差。LIU等[21]研究松木和聚碳酸酯在一定原料比例下共热解行为,发现挥发物-挥发物相互作用促进H2、CO和CO2形成,抑制CnHm生成,主要是因为聚碳酸酯挥发物中碳氢化合物自由基与松木挥发物中含氧自由基之间相互作用生成更多含氧化合物。松木半焦多孔结构和碱(碱土)金属促进聚碳酸酯挥发物中酚类中间体的脱氧和裂解,产生更多CO和H2。为调控煤和生物质热解挥发分的二次反应,提高焦油产率或品质,需进一步研究填料方式对煤和生物质共热解协同作用影响机制。
基于此,笔者利用固定床反应器,用瞬间进料探究填料方式研究淖毛湖煤(NMH)和棉秆(CS)共热解协同效应。通过分析产物分布、焦油品质、焦油组成和半焦孔隙特征,阐述填料方式对共热解协同作用的影响机制,为共热解产物定向调控提供理论基础。
1 试 验
1.1 原料与试剂
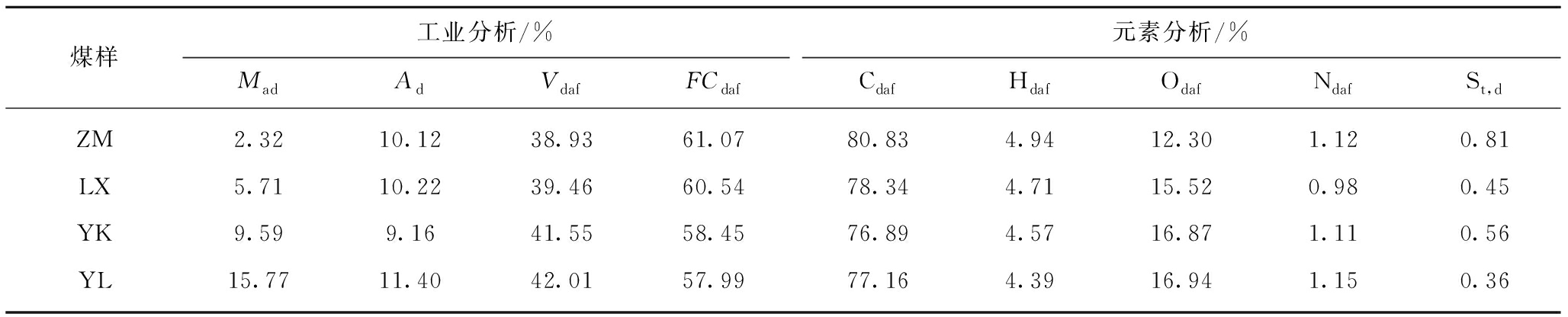
原料为新疆地区淖毛湖煤(NMH)和棉秆(CS)。将原料粉碎、筛分至0.15~0.18 mm,保存备用。NMH和CS工业分析和元素分析见表1。试验所用甲苯购自天津市盛淼精细化工公司,二硫化碳购自天津市鑫铂特化工有限公司,无水硫酸钠购自成都市科隆化学品有限公司,碳酸氢钠购自天津市盛淼精细化工公司。
表1 NMH和CS的工业分析和元素分析
Table 1 Proximate and ultimate analyses of NMH and CS

注:*为差减法。
1.2 热解试验
在固定床反应器上进行淖毛湖煤、棉秆单独热解及共热解试验,装置示意如图1所示。

图1 固定床反应器示意
Fig.1 Schematic diagram of the fixed-bed reactor
反应开始前,先取适量石英棉放入石英吊篮底层用于支撑原料,每次试验原料用量3 g,用铁丝将石英吊篮悬挂在石英管上端。试验开始前用100 mL/min的N2(纯度> 99.999%)吹扫20 min,以排净装置内空气,然后将反应器温度升至600 ℃,用瞬间进料方式,将石英吊篮快速推入恒温区,恒温时间30 min。热解产生的可凝液体产物在-20 ℃下冷凝收集,不可凝气体依次经装有饱和碳酸氢钠的洗气瓶和硅胶干燥管,进行脱硫化氢净化和脱水干燥处理,经干式气体流量计计量气体体积后用铝箔气袋收集。选取煤和棉秆共热解比例为3∶1,所用4种填料方式包括:煤与棉秆分隔放置(Case 1)、煤与棉秆机械混合(Case 2)、煤在棉秆上层(Case 3)和煤在棉秆下层(Case 4),如图2所示。

图2 NMH与CS的填料方式示意
Fig.2 Schematic diagram of filling mode
1.3 产率计算
热解产生的可冷凝液体产物包含热解水和焦油,以甲苯为溶剂分离油水混合物,根据ASTM D 95—2013《蒸馏法测定石油产品和沥青材料中含水量的标准试验方法》方法测定液体产物中水的质量(mwater),由差减法可知焦油质量(mtar)。热解半焦质量(mchar)通过反应前后石英吊篮质量差减得出,NMH半焦和CS半焦分别记为NMH-C和CS-C。采用GC-4000A气相色谱仪(Inficon)。焦油产率(Ytar, %)、水产率(Ywater, %)、半焦产率(Ychar, %)、气体产率(Ygas, %)和气体产量(Yi, mL/g)计算公式如式(1)~(5)所示。
Ytar=mtar/m0,
(1)
Ywater=mwater/m0,
(2)
Ychar=mchar/m0,
(3)
Ygas=(∑MiVi/Vm)/m0,
(4)
Yi=1 000Vi/m0,
(5)
式中,m0为原料质量,g;Mi为各气体组成的摩尔质量,g/mol;Vi为各气体组成的体积,L;Vm为气体(H2、CH4、CO、CO2、C2~C4)摩尔体积,L/mol。
共热解试验中,用产物产率、气体组成、焦油馏分和焦油组成的试验值与计算值差值(ΔY)量化评价NMH与CS共热解协同效应[22-23],见式(6):
ΔY=Yexp-Ycal。
(6)
其中,Yexp和Ycal分别为试验值和计算值,Ycal根据NMH和CS单独热解试验值按质量分数加和计算获得:
Ycal=x1YNMH x2YCS,
(7)
式中,YNMH和YCS分别为NMH和CS单独热解试验值;x1和x2分别为混合物中NMH和CS质量分数。
1.4 产物分析
原料热失重特性在TGA 8000型热重分析仪(美国,PerkinElmer)上测试,称取(10±0.2) mg样品放入坩埚中,在100 mL/min高纯氮气气氛中以10 ℃/min从40 ℃升温至800 ℃。用vario MACRO cube型元素分析仪(德国,Elementar)测定样品中C、H、N和S元素含量,O元素含量由差减法计算得到。焦油馏分组成利用Agilent 7890B气相色谱仪测定,利用ASTM D2887《用气相色谱分析法测定石油馏分沸程分布的标准试验方法》分析焦油品质。测试时初始柱温设定为50 ℃,以10 ℃/min升至300 ℃,保持6 min。根据焦油馏程不同,将焦油分为轻油(< 170 ℃)、酚油(170~<210 ℃)、萘油(210~<230 ℃)、洗油(230~<300 ℃)、蒽油(300~<360 ℃)和沥青(≥360 ℃)[24]。用Agilent 7890B-5977型气相色谱/质谱联用仪(GC/MS)分析焦油组成。具体测试条件为:初始柱温30 ℃,以10 ℃/min升至60 ℃,保持1 min;以3 ℃/min升至90 ℃,保持1 min;以3 ℃/min升至170 ℃,保持1 min;以3 ℃/min升至300 ℃,保持8 min。根据NIST19数据库对各物质质谱图进行解谱,并通过峰面积归一化法计算焦油中各组分相对质量分数。
半焦比表面积和孔径分布等通过ASAP 2460型物理吸附仪(美国,Micromeritics)在液氮温度(-196 ℃)下测定。测试前,将样品在150 ℃条件脱气7 h以除去吸附的气体杂质。并利用氮气等温吸附曲线计算样品分形维数,可定量描述物质表面不规则程度和复杂程度[25]。物质表面越粗糙,孔隙越丰富,则分形维数越大。因此分形维数可定量表征半焦孔隙不规整性和孔结构复杂性[26]。基于氮气等温吸附数据,利用Frenkel-Halsey-Hill(FHH)分形模型计算半焦吸附孔分形维数[27]。FHH模型认为吸附于分形表面上气体可表示为:
ln V=Aln[ln(p0/p)] C,
(8)
A=D-3或A=(D-3)/3。
(9)
其中,V为相对气压下气体吸附量,cm3/g;A为拟合出的线性方程的斜率;p0为吸附气体饱和蒸汽压,Pa;p为系统平衡压强,Pa;C为常数;D为材料分形维数。用2种方法分别计算D,当D在2~3时说明半焦满足孔结构分形特征。
2 结果与讨论
2.1 TG/DTG分析
NMH和CS在氮气气氛下热失重曲线如图3所示。相关参数见表2。由图3(a)可知从室温到150 ℃出现第1个失重阶段,归因于脱除样品孔隙中吸附的水和气体。350~650 ℃是NMH主要失重阶段,在该阶段煤有机大分子结构被破坏,并逸出大量挥发物。DTG曲线最大失重速率位于465 ℃,此时最大失重速率0.279%/℃。第3阶段为650 ℃后,半焦进一步发生缩聚反应生成焦炭。而CS主要热解区间在200~500 ℃(图3(b)),在320 ℃附近,对应最大失重速率0.631%/℃。二者DTG曲线半峰宽相差小,较NMH而言,CS在相同温度区间内失重量61.47%。在室温~800 ℃时,NMH总失重率45.76%,CS总失重率71.20%。

图3 NMH和CS的TG-DTG曲线
Fig.3 TG-DTG curves of NMH and CS
表2 NMH和CS的热重参数
Table 2 Thermogravimetry parameters for NMH and CS

注:Tp为最高峰值温度;ηPTG,max为最大失重速率;ΔT1/2为半峰宽;Δm为总失重量。
CS和NMH热解反应特性差异不仅与二者化学成分有关,更与二者结构组成有关。CS主要是由半纤维素、纤维素和木质素通过多种共价键及氢键相互交联而成的聚合物[28-29]。随着热解温度升高,各组分热解产生的挥发物间会发生交互作用,构成了CS主要热解区间。CS主要化学组成拟合DTG曲线(150~600 ℃)如图4所示,对CS热解区间150~600 ℃的DTG曲线进行分峰拟合。最先热解的是半纤维素,其对应热解区间200~350 ℃,其次是纤维素,280~400 ℃,CS最大失重峰正好在纤维素热解范围内,且在此区间内纤维素热解贡献占比最大。区别于半纤维素及纤维素多糖结构,木质素由对羟基苯基、愈创木酚和丁香基3个结构单元组成。木质素在较宽温度范围(200~550 ℃)内分解,基本涵盖CS整个热解过程[30-31]。

图4 CS主要化学组成拟合DTG曲线(150~600 ℃)
Fig.4 Main chemical composition of CS fits the DTG curves (150-600 ℃)
2.2 产物分布和气体组成
煤与CS质量比为3∶1、热解温度600 ℃,考察4种填料方式下热解产物的收率并分析协同效应,结果如图5所示。由图5(a)和5(b)可知填料方式对热解产物分布影响显著。当用煤在棉秆上层(Case 3)填料方式时,共热解焦油产率明显低于理论计算值,产生负协同效应。用分隔放置(Case 1)、机械混合(Case 2)、煤在棉秆下层(Case 4)填料方式时,焦油产率高于理论计算值,呈现正协同效应。表明这3种填料方式利于NMH与CS热解挥发物间相互作用,CS热解挥发物发挥供氢作用,促进共热解焦油产率提高。由试验结果可知Case 4所得焦油产率最高,为15.94%,较理论计算值增加3.89%,正协同效应最显著。

图5 不同填料方式下NMH与CS共热解产物分布、气体组成和协同效应分析
Fig.5 Products distribution、gas composition and synergistic effect analysis of co-pyrolysis of NMH and CS with varied filling modes
相较Case 2和Case 3,Case 1所得焦油产率明显更高。这是由于Case 2方式下,CS与NMH接触最密集,二者挥发物虽然可及时接触但反应产物未能及时离开恒温区,因此正协同效应较弱。而在Case 3方式下,煤热解产生的挥发物无法及时与下层CS热解产生的富氢挥发物相互作用,且CS半焦的存在延长了煤热解挥发物在恒温区的停留时间,加剧了焦油二次反应,导致焦油产率降低。
此外,CS半焦中AAEMs也催化煤热解挥发物形成轻质芳烃和小分子气态产物[32]。当用Case 1方式时,尽管可一定程度提高焦油产率,但因CS热解产生的富氢挥发物对煤热解挥发物供氢作用有限,焦油产率较Case 4低。对于Case 4方式,CS热解产生的挥发分随载气通过下端NMH层,煤热解挥发物离开煤颗粒表面即可与CS热解产生的富氢组分接触,稳定煤热解产生的大分子自由基碎片,促进热解焦油产率提高。当用Case 1方式时,热解水呈现显著负协同作用,这是由于CS热解生成的·OH等与煤热解挥发物发生相互作用。
为进一步探究焦油产率增加的原因和填料方式对气体产物分布的影响,对热解气体组成定量分析,结果如图5(c)所示。Case 1和Case 3方式时,因热解挥发物在热解中发生缩合反应,导致CH4产量增加。用Case 4方式所得H2、CH4和C2~C4富氢气体产量与理论计算值相比明显降低,这是由于CS热解产生富氢组分参与NMH热解行为,促进焦油生成。
2.3 焦油分析
利用模拟蒸馏对共热解焦油馏分分布分析,结果如表3和图6所示。可知共热解焦油高沸点组分含量(≥360 ℃)均高于低沸点组分(<360 ℃)。Case 4所得焦油中沥青占比最小,为30.23%,轻油占比最高,为26.6%。共热解焦油组分试验值与理论计算值差值如图6所示。可知170 ℃以下沸点焦油组分含量明显减少,而沥青含量大幅增加。对于Case 1、Case 3和Case 4,轻油含量减少,酚油、萘油和洗油含量增加,而蒽油含量减少,这是由于轻油在高温下分解产生较多·H自由基,这些活性自由基利于部分蒽油和沥青裂解,生成酚油、萘油和洗油[33]。在4种填料方式中,Case 2所得焦油中沥青占比最高。表明煤与棉秆共热解对焦油中轻质馏分产生负协同作用,使焦油变得更重。

图6 不同填料方式下共热解焦油馏分协同效应分析
Fig.6 Analysis of synergistic effect of co-pyrolysis tar fraction with varied filling modes
表3 不同填料方式下共热解焦油馏分分布
Table 3 Distribution of co-pyrolysis tar fractions with varied filling modes %

用GC/MS对不同填料方式下共热解焦油组成分析,结果见表4和图7。与理论计算值相比,4种填料方式下焦油中烷烃相对含量均明显增多。主要由于闪速热解使棉秆和煤中挥发分立即释放,大幅缩短相互作用时间跨度。棉秆链烃主链断裂产生大量小分子自由基,这些自由基与煤热解自由基结合,产生更多大分子烃类物质,阻碍碳氢化合物缩合[34]。用Case 2和Case 3方式所得焦油中酚类化合物相对含量分别增加0.70%、4.20%,这主要由于CS分解产生大量含活性氧基团的挥发物与芳烃自由基反应生成稳定的脂肪烃和酚类化合物[35]。研究表明,酚类物质主要来自CS中愈创木酚类物质的二次反应(如脱甲基和脱甲氧基等)[36]。Case 4方式所得焦油中酚类化合物相对含量明显减少的原因是CS热解挥发物与煤热解挥发物发生相互作用,烯烃相对含量增加可归因于共热解中自由基发生了聚合反应[37],而含氧化合物相对含量减少可能因为共热解促进了脱氧反应(如脱羧和脱羰基化等),进一步生成脂肪烃,减少含氧官能团发生交联反应[38]。此外共热解焦油中含氧化合物和N、S杂原子化合物含量均降低,表明富氢自由基与活性含氧基团作用促使焦油中O、N、S原子向固体或气体产物转移[39-40]。

图7 不同填料方式下共热解焦油组分协同效应分析
Fig.7 Analysis of synergistic effect of co-pyrolysis tar components with varied filling modes
表4 不同填料方式下共热解焦油组分相对含量
Table 4 Relative contents of tar components incopyrolysis with varied filling modes %

2.4 半焦分析
2.4.1 孔结构特征
半焦的氮气吸附等温曲线、孔径分布和孔结构参数如图8和表5所示。不同填料方式下煤和棉秆共热解半焦氮气吸附等温线与单独热解半焦类型相同。共热解后,棉秆半焦吸附量减少,而煤半焦的差别较小。由表5可知,与煤热解半焦相比,在不同填料方式下,共热解半焦产生的微孔和介孔明显更多,较大孔隙结构能为二者挥发物间反应提供更多活性位点,利于提高热解转化率[41]。与煤单独热解相比,共热解煤半焦比表面积更大。这是由于共热解过程中,AAEMs促进煤热解反应,使煤半焦表面更粗糙[42]。

图8 半焦N2吸附等温曲线和孔径分布
Fig.8 N2 adsorption isotherm curves and pore size distribution curves of the char
表5 半焦孔结构参数
Table 5 Pore structure parameters of the char

注:a:BET法计算;b:t-plot法计算;c:比表面积-微孔表面积计算;d:在p/p0=0.99时计算得到;e:用BJH法计算。
2.4.2 分形特征分析
基于半焦的氮气吸附等温曲线拟合计算半焦表面结构分形维数,分别计算低压段和高压段分形维数,如图9所示,基于ln V与![]() 线性关系,根据式(9)计算半焦在低压段分形维数D1和高压段分形维数D2,结果见表6。
线性关系,根据式(9)计算半焦在低压段分形维数D1和高压段分形维数D2,结果见表6。

图9 吸附曲线的分形计算结果(FHH模型)
Fig.9 Fractal calculation results of adsorption curves (FHH model)
表6 分形维数计算结果
Table 6 Calcuation results of fractical dimension

低压段吸附力主要依靠范德华力,吸附力大小与半焦表面粗糙度相关;高压段的附主要为毛细凝聚作用,吸附力大小与半焦的结构有关[43]。
由表6可知拟合度R1和R2均接近1,说明拟合度高。用公式D=3 A计算时,分形维数D1和D2均在2~3,表明该方法下半焦粗糙度和孔结构均满足分形结构基本特征。对于Case 3和Case 4方式,位于下层样品半焦的粗糙度变大,这是因共热解中位于上层样品的热解挥发物与位于下层的样品发生相互作用,导致下层样品半焦表面更粗糙。
半焦分形维数D2在2.017~2.700,说明半焦孔结构较复杂。对比不同填料方式下半焦D2值可知用Case 3方式时,下层CS半焦存在会延长煤热解挥发物在恒温区停留时间,CS-C表面出现碳沉积,导致CS原本热解中已打开的孔道被堵上,CS-C分形维数变小。而用Case 4方式时,在共热解中高氧含量CS热解挥发物会促进位于下层NMH的热解转化率,导致下层NMH半焦中原有孔隙变大,且有新的孔隙形成,孔隙数量增加。因此相较单独热解,Case 4方式下共热解会导致位于下层NMH-C的孔隙更不均匀,孔结构更复杂[44]。
3 结 论
1)利用固定床反应器探究4种填料方式对NMH和CS共热解协同效应影响机制。Case 4共热解焦油产率最高,为15.94%,较理论计算值增加3.89%。H2、CH4和C2~C4的产量较理论值降低,表明CS热解产生的富氢组分与NMH热解挥发物发生交互作用,促进焦油生成。
2)Case 2方式所得焦油品质最差。Case 4共热解焦油中沥青占比最小,为30.23%,轻油占比最高,为26.6%。在共热解中,·H自由基与活性含氧基团作用促使焦油中O、N、S原子向固体或气体产物转移。煤与棉秆共热解对焦油中轻油生成产生负协同作用,使焦油变得更重。
3)半焦分形维数D1和D2均在2~3,说明半焦粗糙度和孔结构均满足分形结构基本特征。对于Case 3和Case 4方式,位于下层样品半焦的表面更粗糙。用Case 3方式所得CS-C孔隙更小;而Case 4方式所得NMH-C孔隙更不均匀,孔结构更复杂。
[1] WANG M, FU W, WANG H, et al. Structural changes and prod-uct distribution of typical Xinjiang coals and chars during pyrolysis [J]. The Canadian Journal of Chemical Engineering, 2024, 102(4): 1431-1440.
[2] CHANG H, ZHANG Z, QIANG L, et al. Study on the pyrolysis characteristics of a typical low rank coal with hydrothermal pretreatment [J]. Energy &Fuels, 2019, 33(5): 3871-3880.
[3] WU B, GUO X, LIU B, et al. Insight into hydrogen migration and redistribution characteristics during co-pyrolysis of coal and polystyrene [J]. Journal of Analytical and Applied Pyrolysis, 2023, 173: 106071.
[4] 卢梦仟, 李珊, 石运固, 等. 褐煤及其低温热解半焦中可溶多环芳烃特征 [J]. 洁净煤技术, 2023, 29(11): 58-65.
LU Mengqian, LI Shan, SHI Yungu, et al. Characterization of soluble polycyclic aromatic hydrocarbons in lignite and its low-temperature pyrolysis semi-coke [J]. Clean Coal Technology, 2023, 29(11): 58-65.
[5] 孟德昌, 赵渊, 张亚青, 等. 煤焦油裂解催化剂制备及催化裂解特性 [J]. 洁净煤技术, 2023, 29(2): 139-148.
MENG Dechang, ZHAO Yuan, ZHANG Yaqing, et al. Preparation and catalytic cracking characteristics of coal tar cracking catalyst [J]. Clean Coal Technology, 2023, 29(2): 139-148.
[6] REN X, CAO J, MA M, et al. Insight on product regula-tion and catalysts deactivation during catalytic fast pyrolysis of lignite over micro/mesoporous catalysts synthesized by ZSM-5 seed [J]. Journal of Analytical and Applied Pyrolysis, 2024, 179: 106487.
[7] REN X, ZHAO S, CAO J, et al. Effect of coal ranks on light aromatics production during reforming of pyrolysis volatiles over HZSM-5 under Ar and H2-assisted atmospheres [J]. Journal of Analytical and Applied Pyrolysis, 2020, 152: 104958.
[8] WU L, GUAN Y, LI C, et al. Insight into product characteristics from microwave co-pyrolysis of low-rank coal and corncob: Unraveling the effects of metal catalysts [J]. Fuel, 2023, 342: 127860.
[9] WU Y, ZHU J, YANG J, et al. Insight into co-pyrolysis interaction of Pingshuo coal and low-density polyethylene under varied mixing configurations via in situ Py-TOF-MS [J]. Journal of Analytical and Applied Pyrolysis, 2022, 168: 105698.
[10] KASARP, SHARMA D K, AHMARUZZAMAN M. Thermal and catalytic decomposition of waste plastics and its co-processing with petroleum residue through pyrolysis process [J]. Journal of Cleaner Production, 2020, 265: 121639.
[11] WEN Y, LIU S, FU S, et al. Insight into influence of process parameters on co-pyrolysis interaction between Yulin coal and waste tire via rapid infrared heating [J]. Fuel, 2023, 337: 127161.
[12] LI S, LI J, XU J. Investigating the release behavior of biomass and coal during the co-pyrolysis process [J]. International Journal of Hydrogen Energy, 2021, 46(70): 34652-34662.
[13] LI Y, HUANG S, WANG Q, et al. Hydrogen transfer route and interaction mechanism during co-pyrolysis of Xilinhot lignite and rice husk [J]. Fuel Processing Technology, 2019, 192: 13-20.
[14] HE W, YIN G, ZHAO Y, et al. Interactions between free radicals during co-pyrolysis of lignite and biomass [J]. Fuel, 2021, 302: 121098.
[15] GUO X, LIU Z, XIAO Y, et al. The Boltzmann-Monte-Carlo-Percolation (BMCP) model on pyrolysis of coal: The volatiles′ reactions [J]. Fuel, 2018, 230: 18-26.
[16] YUAN T, HE W, YIN G, et al. Comparison of bio-chars formation derived from fast and slow pyrolysis of walnut shell [J]. Fuel, 2020, 261: 116450.
[17] TIAN B, ZHAO W, GUO Q, et al. A comprehensive understanding of synergetic effect and volatile interaction mechanisms during co-pyrolysis of rice husk and different rank coals [J]. Energy, 2022, 254: 124388.
[18] LI D, LEI S, RAJPUT G, et al. Study on the co-pyrolysis of waste tires and plastics [J]. Energy, 2021, 226: 120381.
[19] LI J, ZHU J, HU H, et al. Co-pyrolysis of Baiyinhua lignite and pine in an infrared-heated fixed bed to improve tar yield [J]. Fuel, 2020, 272: 117739.
[20] ZHU J, ZHAO S, WEI B, et al. Enhanced co-pyrolysis syner-gies between cedar and Naomaohu coal volatiles for tar production [J]. Journal of Analytical and Applied Pyrolysis, 2021, 160: 105355.
[21] LIU X, BURRA K R G, WANG Z, et al. Towards enhanced understanding of synergistic effects in co-pyrolysis of pinewood and polycarbonate [J]. Applied Energy, 2021, 289: 116662.
[22] WU Z, WANG S, ZHAO J, et al. Thermochemical behavior and char morphology analysis of blended bituminous coal and lignocellulosic biomass model compound co-pyrolysis: Effects of cellulose and carboxymethylcellulose sodium [J]. Fuel, 2016, 171: 65-73.
[23] WU Z, LI Y, XU D, et al. Co-pyrolysis of lignocellulosic biomass with low-quality coal: Optimal design and synergistic effect from gaseous products distribution [J]. Fuel, 2019, 236: 43-54.
[24] WEI B, JIN L, WANG D, et al. Catalytic upgrading of lignite pyrolysis volatiles over modified HY zeolites [J]. Fuel, 2020, 259: 116234.
[25] 王然宇. 生物质和烟煤热解/气化机理及其协同作用研究 [D]. 大连: 大连理工大学, 2022.
[26] WU Z, MA C, JIANG Z, et al. Structure evolution and gasification characteristic analysis on co-pyrolysis char from lignocellulosic biomass and two ranks of coal: Effect of wheat straw [J]. Fuel, 2019, 239: 180-190.
[27] ZHU J, LIU J, YANG Y, et al. Fractal characteristics of pore structures in 13 coal specimens: Relationship among fractal dimension, pore structure parameter, and slurry ability of coal [J]. Fuel Processing Technology, 2016, 149: 256-267.
[28] CHEN Y, FANG Y, YANG H, et al. Effect of volatiles interaction during pyrolysis of cellulose, hemicellulose, and lignin at different temperatures [J]. Fuel, 2019, 248: 1-7.
[29] 宋瑞珍, 杨晓阳, 张鹏, 等. 低阶煤和生物质水热碳化特性及水热炭功能化改性研究进展 [J]. 洁净煤技术, 2024, 30(3): 72-85.
SONG Ruizhen, YANG Xiaoyang, ZHANG Peng, et al. Research progress on the hydrothermal carbonization characteris-tics of low rank coal and biomass and the functionalization modification of hydrochar [J]. Clean Coal Technology, 2024, 30(3): 72-85.
[30] LV G, WU S. Analytical pyrolysis studies of corn stalk and its three main components by TG-MS and Py-GC/MS [J]. Journal of Analytical and Applied Pyrolysis, 2012, 97: 11-18.
[31] GE L, ZHAO C, ZUO M, et al. Effects of Fe addition on pyrolysis characteristics of lignin, cellulose and hemicellulose [J]. Journal of the Energy Institute, 2023, 107: 101177.
[32] ZHAO H, SONG Q, LIU S, et al. Study on catalytic co-pyroly-sis of physical mixture/staged pyrolysis characteristics of lignite and straw over an catalytic beds of char and its mechanism [J]. Energy Conversion and Management, 2018, 161: 13-26.
[33] 张君涛, 石润坤, 牛犇, 等. CH4气氛对煤中低温热解焦油产率和质量影响[J]. 煤炭学报, 2021, 46(1): 292-299.
ZHANG Juntao, SHI Runkun, NIU Ben, et al. Effect of CH4 atmosphere on tar yield and quality in coal pyrolysis at low-medium pyrolysis temperature[J]. Journal of China Coal Society, 2021, 46(1): 292-299.
[34] LI M, LU Y, HU E, et al. Fast co-pyrolysis characteristics of high-alkali coal and polyethylene using infrared rapid heating [J]. Energy, 2023, 266: 126635.
[35] MAO F, WANG J, FAN H. Application of two-dimensional gas chromatography/time-of-flight mass spectrometry (GC×GC-TOFMS) for the thorough study of hydrocarbons in lignite pyrolysates [J]. Journal of Analytical and Applied Pyrolysis, 2021, 157: 105178.
[36] ZHU J, JIN L, LI J, et al. Fast pyrolysis behaviors of cedar in an infrared-heated fixed-bed reactor [J]. Bioresource Technology, 2019, 290: 121739.
[37] SHI Z, JIN L, ZHOU Y, et al. In-situ analysis of catalytic pyrolysis of Baiyinhua coal with pyrolysis time-of-flight mass spectrometry [J]. Fuel, 2018, 227: 386-393.
[38] YANG Q, YAO Q, MA D, et al. Investigation of the (cataly-tic) co-pyrolysis of Shendong coal and coal tar based on rapid pyrolysis and ANN modelling [J]. Journal of Analytical and Applied Pyrolysis, 2022, 163: 105486.
[39] WU L, LIU J, ZHOU J, et al. Evaluation of tar from the microwave co-pyrolysis of low-rank coal and corncob using orthogonal-test-based grey relational analysis (GRA) [J]. Journal of Cleaner Production, 2022, 337: 130362.
[40] LIU J, ZHU Y, MIAO Z, et al. Study on the pretreatment pr-ocess and removal rules of sulfur-containing compounds for medium- and low-temperature coal tar [J]. ACS Omega, 2021, 6(19): 12541-12550.
[41] ZHANG X, BAI Y, WEI J, et al. Study on char-ash-slag-liquid transition and its effect on char reactivity [J]. Energy &Fuels, 2020, 34(3): 3941-3951.
[42] MENG H, WANG M, WU Z, et al. Co-pyrolysis of platanus wood and bituminous coal: Product distributions, char pore analysis and synergistic effects [J]. Journal of Analytical and Applied Pyrolysis, 2022, 167: 105703.
[43] 李子文. 低阶煤的微观结构特征及其对瓦斯吸附解吸的控制机理研究 [D]. 徐州: 中国矿业大学, 2015.
[44] WU Z, YANG W, MENG H, et al. Physicochemical structure and gasification reactivity of co-pyrolysis char from two kinds of coal blended with lignocellulosic biomass: Effects of the carboxymethylcellulose sodium [J]. Applied Energy, 2017, 207: 96-106.
Synergetic effect during the co-pyrolysis of Naomaohu coal and cotton stalk

移动阅读